In intact cells, however, mitochondria are exposed to metabolic and environmental fluctuations, interact with other organelles, and receive inputs from cell signaling pathways. The chemiosmotic theory is rooted in measurements of bioenergetics parameters, such as oxygen consumption, ATP production, pH, and membrane potential, in isolated mitochondria artificially maintained under different metabolic conditions. 
The 13 mitochondrial-encoded proteins include seven subunits of the respiratory chain complex I, one of complex III, three of complex IV, and two of the complex V, i.e., all the respiratory chain complexes that are involved in the transport of protons. Mitochondria are endosymbiotic organelles, and virtually all of the ∼1,500 genes required to build a functional mitochondria have been transferred to the chromosomes of the host cell, except for those coding for 13 polypeptides of the OXPHOS subunits, plus the ribosomal and transfer RNAs required for their synthesis. The importance of mitochondrial proton transport is highlighted by the retention of genes coding for OXPHOS subunits within the mitochondrial genome. The energy stored in the proton gradient is then used to drive the activity of the ATP synthase (complex V) that catalyzes the conversion of ADP to ATP within the mitochondrial matrix. According to the chemiosmotic theory first postulated by Mitchell (1975), the free energy (ΔG) released by the oxidation of highly reduced energetic substrates is used by the complexes I, III, and IV of the electron transport chain to generate a proton gradient across the IMM. All of these functions rely on the ability of mitochondria to move protons across their inner membrane during oxidative phosphorylation (OXPHOS), the process that couples the oxidation of energetic substrates to the synthesis of ATP. Mitochondria are multifunctional organelles involved in energy conversion, lipid metabolism, heat production, Ca 2+ signaling, reactive oxygen species (ROS) production, and apoptosis. Unlike the “superoxide flashes” reported with a pH-sensitive circularly permuted YFP (cpYFP), these “pH flashes” preserve the Δp during spontaneous fluctuations in ΔΨ m therefore, we propose that the flashes are energy conservation events that reflect the intrinsic properties of the mitochondrial proton circuit. These measurements have revealed that the pH mito is in dynamic equilibrium with the cytosolic pH and that spontaneous pH mito elevations coinciding with ΔΨ m drops occur in single mitochondria. 
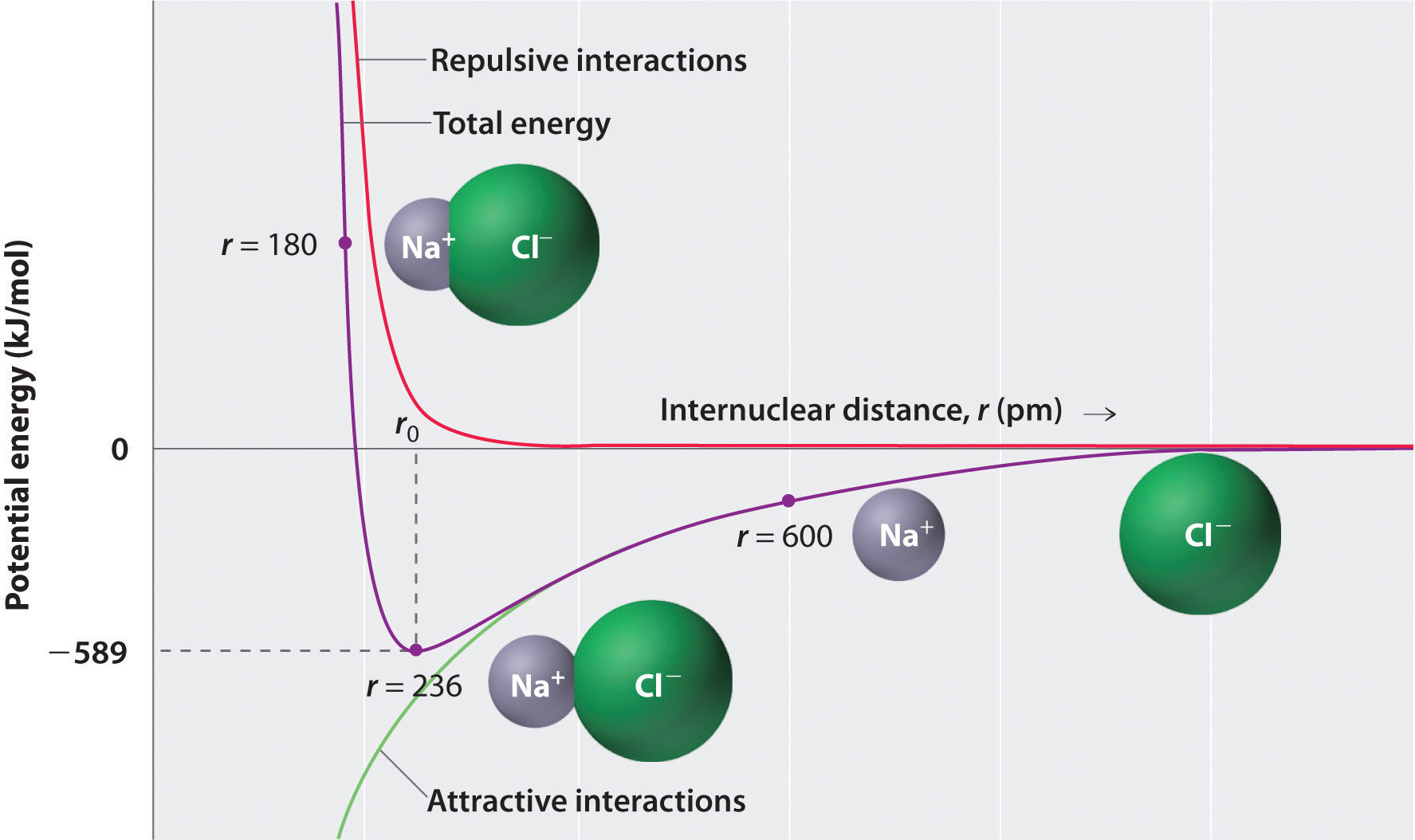
In this Perspective, we discuss the putative roles of the pH mito and review the different techniques used to measure pH mito and ΔpH m in isolated mitochondria and in intact cells, focusing on our recent results obtained with genetically encoded pH-sensitive indicators. ΔpH m, however, drives the fluxes of metabolic substrates required for mitochondrial respiration and the activity of electroneutral ion exchangers that maintain mitochondria osmolarity and volume, and recent studies indicate that the mitochondrial pH (pH mito) plays an important and underappreciated role in physiological and pathological situations such as apoptosis, neurotransmission, and insulin secretion. Because ΔΨ m contributes most of the Δp and can be easily measured in intact cells with fluorescent dyes, most studies ignore the contribution of ΔpH m and only record changes in ΔΨ m to track the metabolic state of mitochondria. Studies in isolated mitochondria have established that the chemical gradient for protons (ΔpH m) and the mitochondrial membrane potential (ΔΨ m) contribute independently to the proton-motive force (Δp) that drives the synthesis of ATP. On the addition of substrate, O 2 is consumed in electron transport so levels O 2 levels fall linearly.The generation of a proton gradient across the inner mitochondrial membrane (IMM) is an essential energy conservation event that couples the oxidation of carbohydrates and fat to the synthesis of ATP. Until substrate is added, O 2 levels are constant. The respiration rate is given by the slope of the curve. Panel ( B) shows changes in O 2 with time. Note that this step allows the regeneration of ADP and efficiency can be measured at any ADP concentration. Panel ( A) shows the chemistry involved in the determination of the ATP/O ratio to measure coupling efficiency. \): Measurement of coupling efficiency in respiring mitochondria. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
